STROKE
 Ischemic stroke is one of the most frequent causes of death and the leading cause of disability in adults worldwide. It results from obstruction of one or more cerebral arteries causing loss of blood flow in respective brain regions and in consequence loss of neurological function and brain tissue. Ischemic deterioration continues over hours or even days and depends primarily on the severity and the duration of ischemia. In core regions of the ischemic territory, injury is rapid and soon irreversible. Functionally impaired but structurally intact tissue between core and healthy brain is termed ischemic penumbra. It is this region where a cascade of neurochemical and neurophysiological processes contribute to progressive brain damage. Numerous experimental studies have demonstrated that most of these processes can be counteracted by therapeutic neuroprotective intervention. Effective translation into clinical practice, however, has failed up to now. The only approved treatment of stroke is still early recanalization and reperfusion by intravenous recombinant tissue plasminogen activator (rt-PA) or mechanical thrombectomy with stent retrievers.
Ischemic stroke is one of the most frequent causes of death and the leading cause of disability in adults worldwide. It results from obstruction of one or more cerebral arteries causing loss of blood flow in respective brain regions and in consequence loss of neurological function and brain tissue. Ischemic deterioration continues over hours or even days and depends primarily on the severity and the duration of ischemia. In core regions of the ischemic territory, injury is rapid and soon irreversible. Functionally impaired but structurally intact tissue between core and healthy brain is termed ischemic penumbra. It is this region where a cascade of neurochemical and neurophysiological processes contribute to progressive brain damage. Numerous experimental studies have demonstrated that most of these processes can be counteracted by therapeutic neuroprotective intervention. Effective translation into clinical practice, however, has failed up to now. The only approved treatment of stroke is still early recanalization and reperfusion by intravenous recombinant tissue plasminogen activator (rt-PA) or mechanical thrombectomy with stent retrievers.
Several reasons are thought to be responsible for this failure of translation. Clinical studies for example tend to include, unlike experimental studies, rather heterogeneous groups of patients. Clinical studies normally begin neuroprotective treatment rather late after stroke onset, whereas experimental studies start very early or even before arterial occlusion. Therefore, adequate monitoring should more specifically identify pathophysiological mechanisms to identify target patient populations prone to more specific therapeutic interventions. Such individualized approach would also allow better temporal matching of therapeutic interventions. On this basis, neuroprotection might be transformed more successfully into clinical practice.
Cortical Spreading Depolarization (CSD) is a pathophysiological mechanism that has been identified in the ischemic penumbra of many experimental stroke models and has consequently been termed Peri-Infarct Depolarization (PID). CSDs/PIDs propagate across cerebral cortex or in more general terms across cerebral grey matter. They are characterized by almost complete ion homeostasis failure of neurons as well as glial cells. Experimental studies show that they put an enormous energetic burden on tissue in the ischemic penumbra, which causes expansion of the ischemic core. Another mechanism that augments secondary deterioration is the failure or even the inversion of the typical blood flow raise coupled to CSD/PID. Under physiological conditions, this response would compensate for the increased demand of energy substrates, i.e., glucose and oxygen.
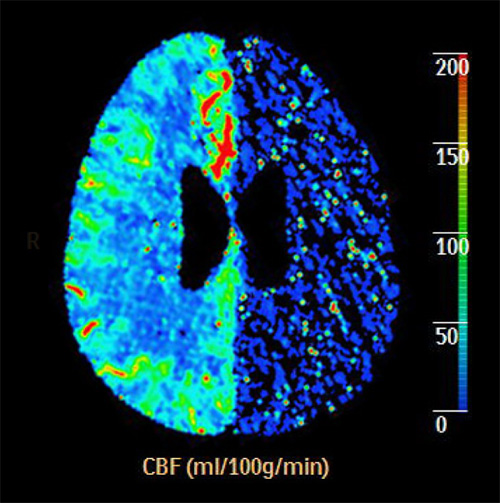
A first clinical study of the COSBID group on ischemic stroke has been performed in malignant hemispheric stroke (MHS) patients. MHS comprises around 10% of stroke patients. These patients have massive infarction (see figure) including a large part of the territory of the middle cerebral artery (MCA) and often additionally that of neighboring arteries. This type of stroke requires decompressive craniectomy to avoid life-threatening brain edema and resulting elevation of intracranial pressure. Because of the craniectomy, access to cortex is provided and allows subdural electroencephalography, a requirement to detect CSD/PID. In the first study on MHS patients, we have detected a very high incidence of CSD/PID occurrence, and have now preliminary evidence for infarct progression after stroke onset. In the ongoing investigations, we examine the relationship between CSD/PID occurrence and infarct progression using sequential MR Imaging, and aim to correlate CSD/PID occurrence and infarct progression to neurological outcome. Our studies on MHS patients will provide the basis for work on other groups of patients suffering from ischemic stroke as soon as non-invasive methods for the detection CSD/PID are available, and will provide the basis for individualized and therefore hopefully more successful therapeutic approaches.
